
Taking a new drug to a foreign market takes more than just proving it works. You have to translate those clinical outcomes into the language of local regulators and health economists.
This case study looks at what it takes to bring the antipsychotic drug KarXT to the English market after its FDA approval in the US. I built a knowledge graph to map KarXT’s safety profile and efficacy data from the EMERGENT-3 trial straight to UK regulatory guidelines.
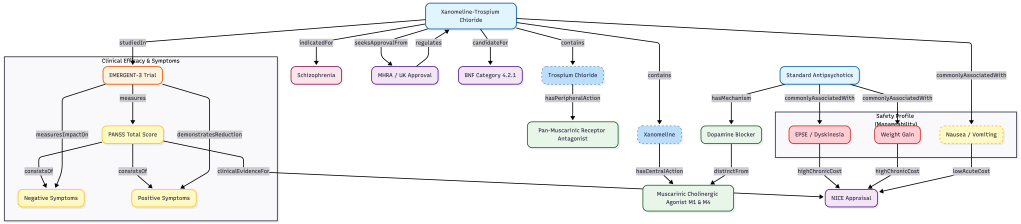
Moving beyond basic taxonomy
The main challenge in pharmaceutical data architecture is interoperability. Instead of just making a flat list of drug attributes, I designed a relational ontology that links mechanisms of action directly to symptom reduction and downstream economic impacts.
Here is why this model works well.
- I deconstructed the molecule for better querying. Instead of just tagging KarXT as an antipsychotic, I modeled its two distinct components. Xanomeline acts as the central nervous system agonist, while Trospium works as the peripheral nervous system antagonist. This lets data scientists query specifically for peripheral side effect mitigation, which is a massive differentiator from standard care.
- I tracked unmet clinical needs at a granular level. The trial data showed different efficacy signals for positive and negative symptoms, so I split the PANSS scores into separate nodes. Separating them opens the door for future analysis on treatment-resistant negative symptoms, a high-value unmet need.
- I translated safety into financial viability. I explicitly linked adverse events like weight gain to economic value relationships. A graph query can now traverse from the drug’s components to its side effect profile, bypassing the severe side effects of standard drugs, and going straight into NICE appraisal logic. This turns safety data into a direct financial argument.
- I ensured UK regulatory interoperability. To make sure the data is actually useful in the UK, I aligned the ontology classes with BNF categories and MHRA regulatory pathways. When this data feeds into real-time analytics, it already speaks the language of the UK health system, completely reducing the risk of regulatory misalignment.
Mapping the ontology classes
To stay perfectly aligned with UK standards, I mapped the data to established taxonomies.
| Ontology class (concept) | Data | UK standard / taxonomy |
| Active substance | Xanomeline, Trospium Chloride | dm+d (dictionary of medicines & devices) |
| Mechanism of action | Muscarinic M1/M4 agonist, pan-muscarinic receptors antagonist | SNOMED CT (observable entity) |
| Clinical trial | EMERGENT-3 trial (NCT04738123) | HRA (Health Research Authority) standards |
| Clinical outcome | PANSS total score, CGI-S | NICE appraisal criteria |
| Adverse event | Dyspepsia, vomiting, GERD | MedDRA, BNF |
| Regulatory body | FDA (implied, already reached), MHRA (target) | MHRA |
| Comparator | Standard antipsychotics (dopamine blockers) | BNF (antipsychotic drugs) |
Interrogating the graph with Cypher
Because the data is structured in a Neo4j graph database, we can pull complex insights with simple queries.
If we want to find drugs, their associated side effects, and the resulting cost impact on a NICE appraisal
Cypher
MATCH (d:Drug)-[:COMMONLY_ASSOCIATED_WITH]->(safetyNode)-[costImpact]->(n:Regulatory {name: 'NICE Appraisal'}) [cite: 34]RETURN [cite: 35]d.name AS Drug, [cite: 36]labels(safetyNode)[0] AS SafetyCategory, [cite: 37]safetyNode.name AS SideEffect, [cite: 38]type(costImpact) AS EconomicImpactToNICE [cite: 39]
Or if we want to show the distinct components of KarXT and their respective mechanisms
Cypher
MATCH (karxt:Drug {id: 'KarXT'})-[:CONTAINS]->(comp:Component)-[action]->(mech:Mechanism) RETURN karxt.name AS Formulation, comp.name AS ActiveIngredient, type(action) AS ActionPathway, mech.name AS ReceptorTarget [cite: 41]
Wrapping up
Good data architecture does more than store information. It tells a story. By thoughtfully designing this ontology, the clinical achievements of the EMERGENT-3 trial are mapped to prove actual medical and economic value to UK regulators.
(Source data from Kaul, I., et al. (2024). Efficacy and Safety of Xanomeline-Trospium Chloride in Schizophrenia: A Randomized Clinical Trial (EMERGENT-3). JAMA Psychiatry.)